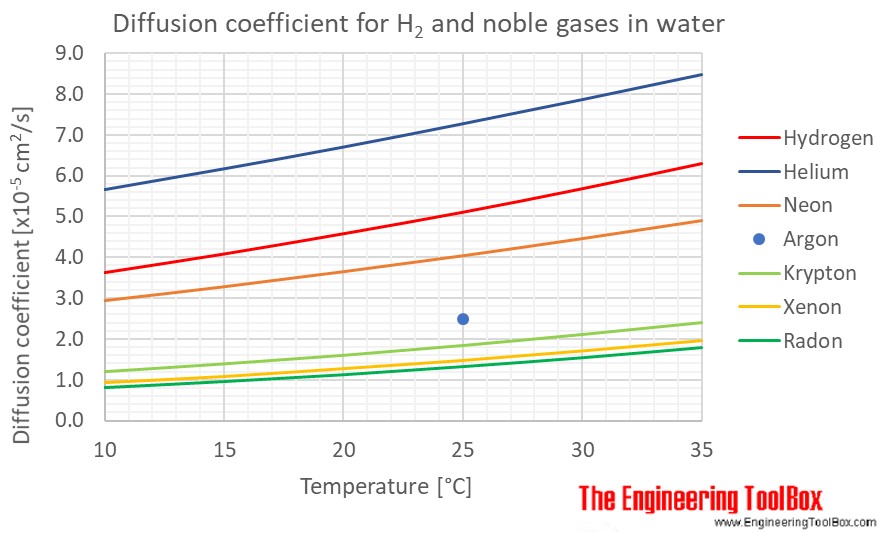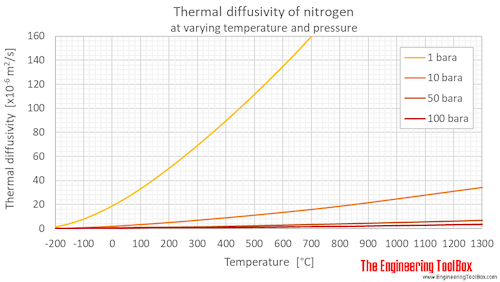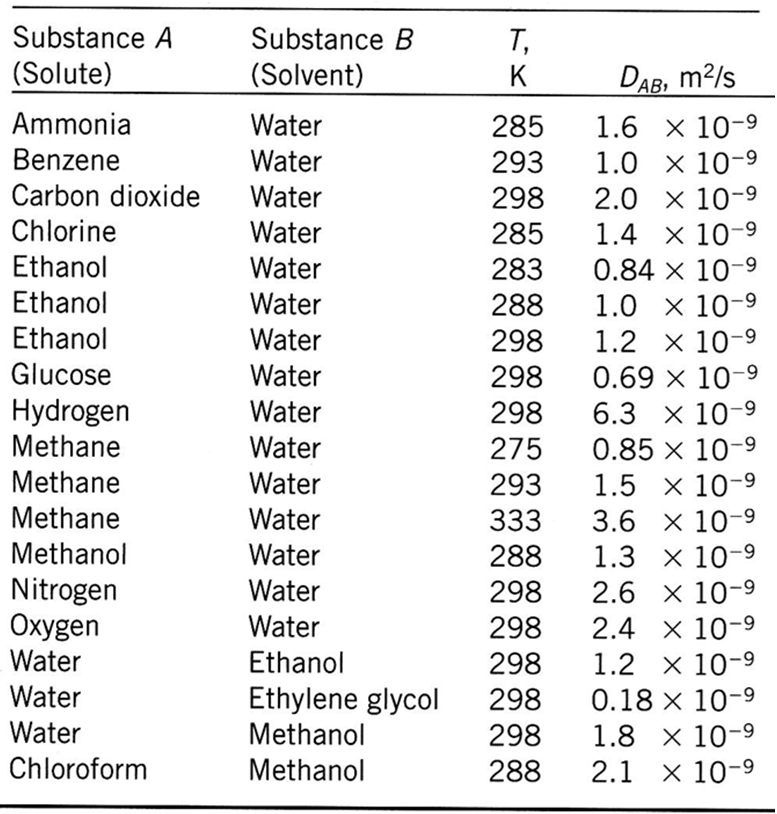
Exercise: Binary diffusion coefficients of some liquid mixtures — Introduction to Chemical and Biological Engineering
![PDF] Solubilities and diffusion coefficients of carbon dioxide and nitrogen in polypropylene, high-density polyethylene, and polystyrene under high pressures and temperatures | Semantic Scholar PDF] Solubilities and diffusion coefficients of carbon dioxide and nitrogen in polypropylene, high-density polyethylene, and polystyrene under high pressures and temperatures | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1e7956c9de5cf024494a6948054c30b18384260b/5-Table1-1.png)
PDF] Solubilities and diffusion coefficients of carbon dioxide and nitrogen in polypropylene, high-density polyethylene, and polystyrene under high pressures and temperatures | Semantic Scholar

Metals | Free Full-Text | The Anisotropic Stress-Induced Diffusion and Trapping of Nitrogen in Austenitic Stainless Steel during Nitriding
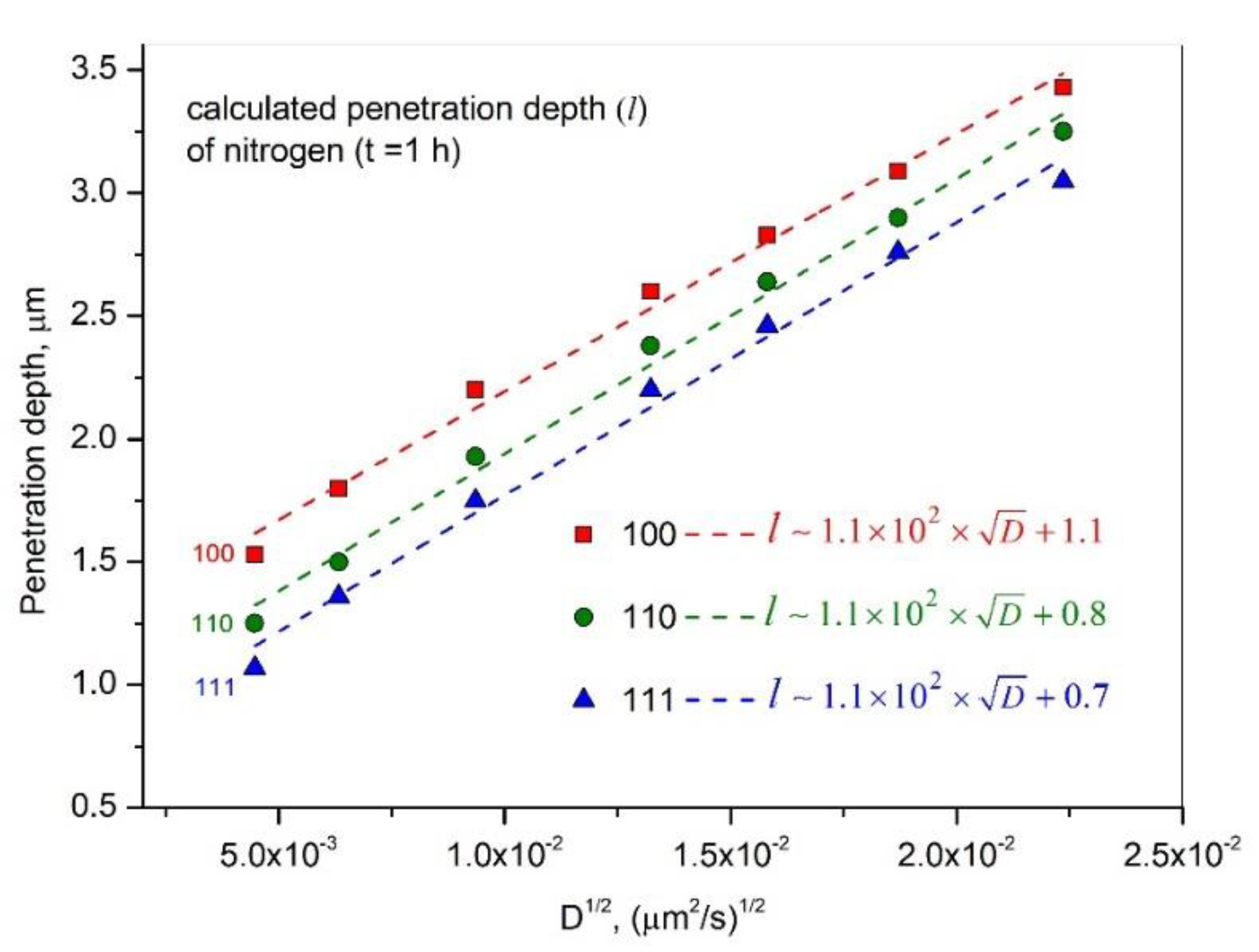
Metals | Free Full-Text | The Anisotropic Stress-Induced Diffusion and Trapping of Nitrogen in Austenitic Stainless Steel during Nitriding

Color online) Diffusion coefficients of hydrogen (D H ), nitrogen (D N... | Download Scientific Diagram

Literature values for nitrogen diffusion in stainless steel (Mändl et... | Download Scientific Diagram

Table 2 from Determination of the oxygen and nitrogen interstitial diffusion coeficient in niobium by mechanical spectroscopy | Semantic Scholar

Diffusion Coefficients of CO2 and N2 in Water at Temperatures between 298.15 K and 423.15 K at Pressures up to 45 MPa | Journal of Chemical & Engineering Data

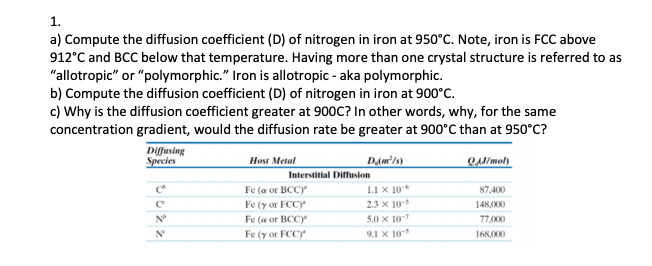
SOLVED: 5. Compare the diffusion coefficients for hydrogen and nitrogen in FCC iron at 1000°C and explain the difference in their values. Diffusion Couple Q (cal/mol) - Q is the activation energy

Binary Diffusion Coefficients for Methane and Fluoromethanes in Nitrogen | Journal of Chemical & Engineering Data
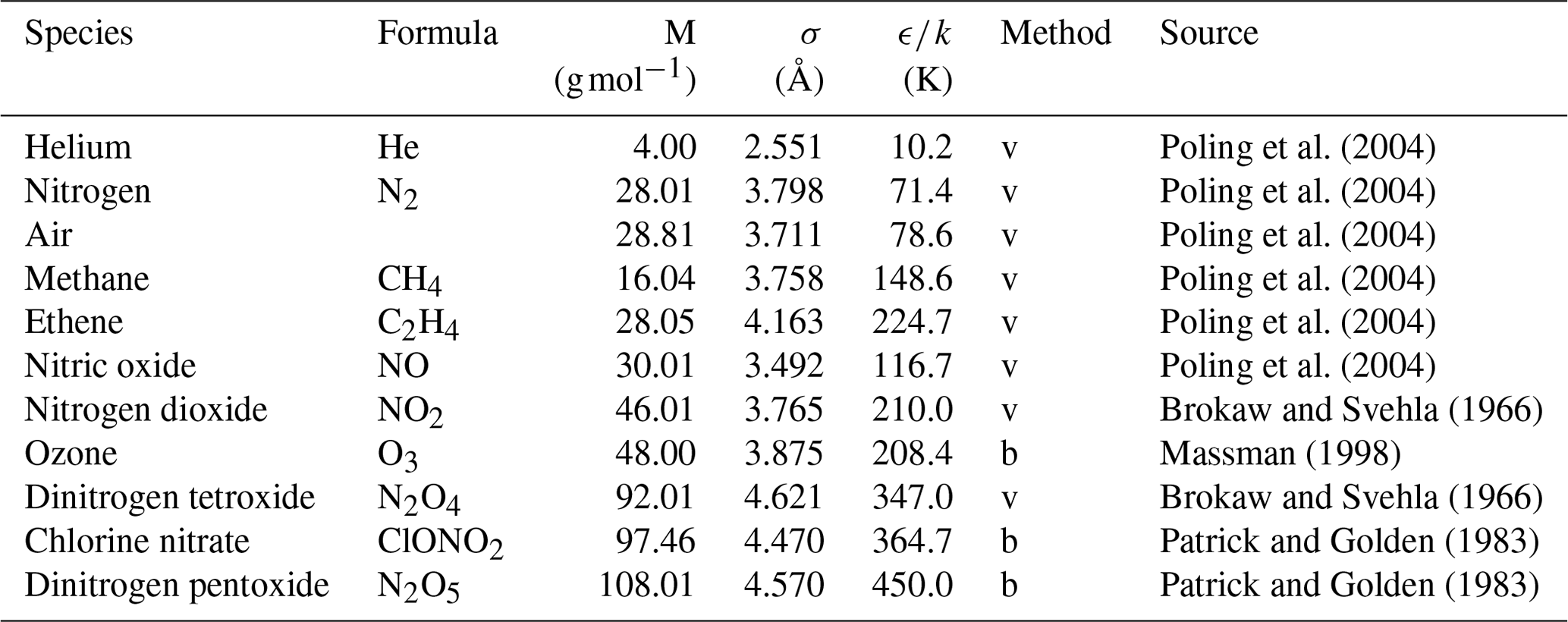
ACP - Technical note: Determination of binary gas-phase diffusion coefficients of unstable and adsorbing atmospheric trace gases at low temperature – arrested flow and twin tube method




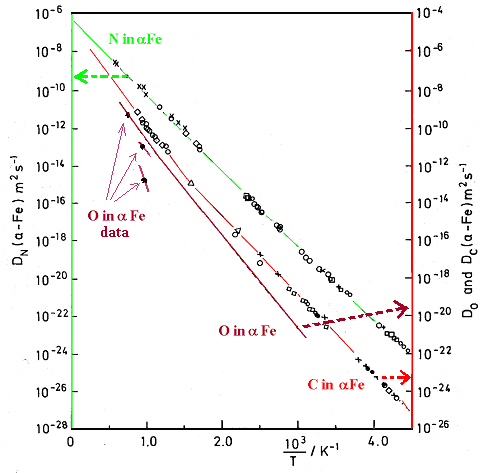

![Nitrogen diffusion coefficient in the Fe-Ni system at 1600 C. [14] | Download Scientific Diagram Nitrogen diffusion coefficient in the Fe-Ni system at 1600 C. [14] | Download Scientific Diagram](https://www.researchgate.net/publication/227114518/figure/fig3/AS:393846249803779@1470911671918/Nitrogen-diffusion-coefficient-in-the-Fe-Ni-system-at-1600-C-14.png)